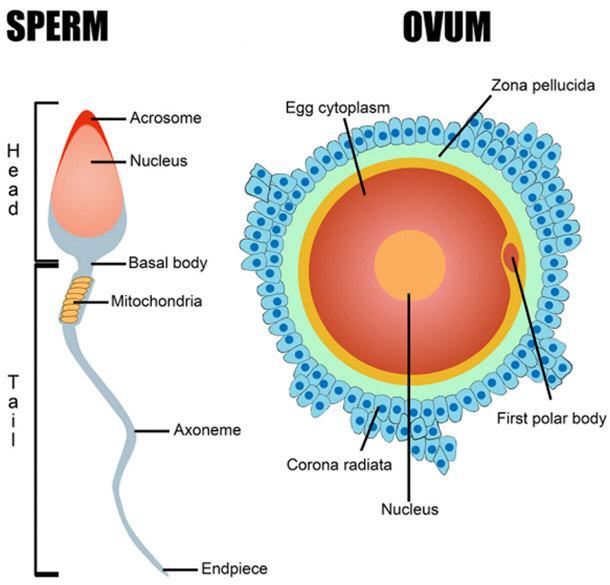
FLC differentiation is thought to be influenced by SC activity, and it has been demonstrated that SC products like desert hedgehog (DHH) or platelet derived growth factor (PDGFA) are essential for FLC development. However, none of the suggested progenitor pools have been shown to directly give rise to FLCs. Evidence also suggests that FLCs and adrenal cortex cells share a common ancestor. In rodents, FLCs are found in the testicular interstitium on gestational days 11–12 and are assumed to derive from numerous embryonic tissues, including the coelomic epithelium, migrating neural crest cells and mesonephric or epithelial populations of the neighboring mesonephros, early after testis determination. The present review discusses recent insights from studies of the formation and function of LCs, as well as recent breakthroughs in understanding LCs contribution to diminished testicular function in spermatogenic failure.Īlthough the origin of FLCs is unknown, several hypotheses have emerged. To understand early susceptibilities to disturbance, we must first understand T production, including the cells involved. Subsequently, it has been proposed that there is a linkage between male reproductive disorders that occur at birth and those that emerge in adolescence. In rats, experimentally-induced testicle disorders occur in response to disruption of fetal androgen production/action during MPW. Proper masculinization requires adequate testosterone (T) production early in fetal development, specifically in the masculinization programming window (MPW). Variations in androgen levels may be significant signs of either causal or developmental disturbances of both the androgen-producing and spermatogenic compartments of the human testicle. These changes worsen with the severity of testis dysfunction. Such deteriorated functioning is mainly manifested as histological and hormonal alterations in the testis compartment. Spermatogenic failure, manifested as azoospermia, is frequently linked to impaired LC activity. In addition, LCs produce proteins with endogenous and xenotoxic metabolic functions, which also reduce oxidative stress, which thus protects the testicle from toxins. LCs, on the other hand, are in charge of the synthesis and secretion of androgens, necessary for the masculinization of male fetuses, together with the initiation and maintenance of spermatogenesis. SCs are responsible for supporting the regulation of the spermatogenesis process and the supply of nutrients to germ cells. The main cells engaged in maintaining these functions are Leydig cells (LCs), which reside in the testicular intertubular space, and Sertoli cells (SCs), which dwell inside seminiferous tubules. 
Not only are these functions fundamental for the upkeep of male sexual characteristics, but they are also fundamental for fertility and species preservation. 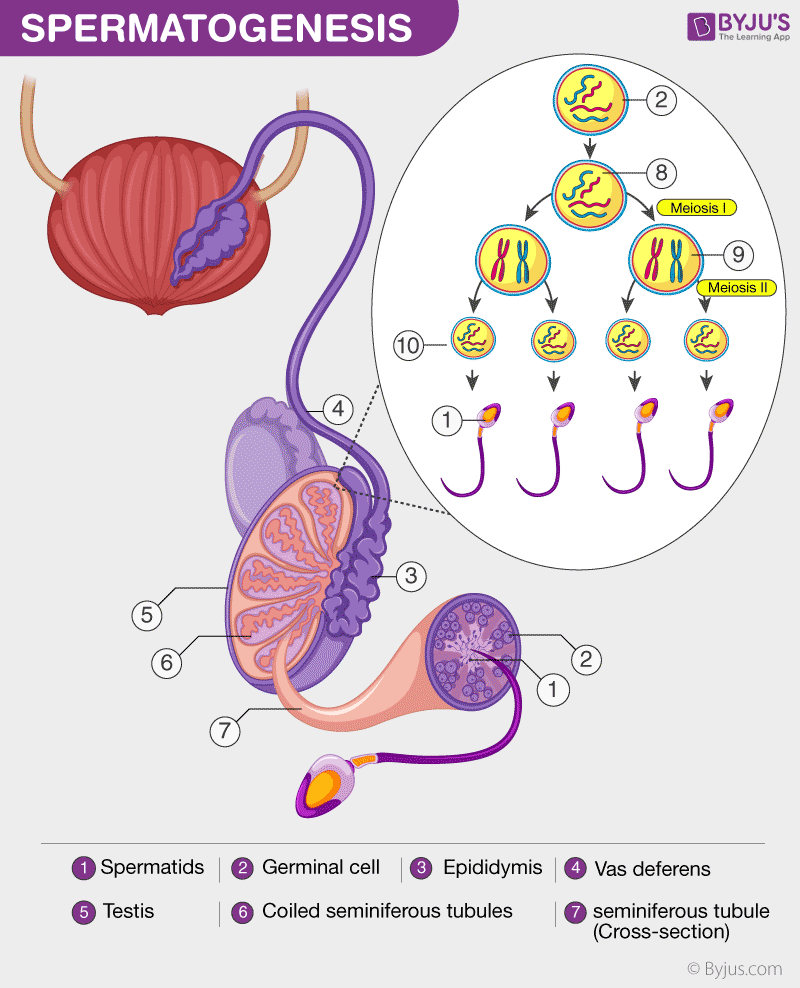
The testicle is the primary organ responsible for maintaining male fertility and the hormonal state.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
